Current Projects
|
|
Techniques
X-ray diffraction, scanning electron microscopy coupled with
electron-dispersive X-ray microscopy, electron microprobe analysis,
Fourier transform infrared spectroscopy,
time-resolved X-ray diffraction coupled with flow-through
reactions, inductively coupled plasma mass spectrometry, X-ray
absorption spectroscopy,
Rietveld analysis, reactive transport modeling, synchrotron X-ray fluorescence mapping
Publications
Kim, JJ, Ling, FT, Platternberger, DA, Clarens, AF, Lanzirotti, A, Newville, M, Peters, CA. “SMART mineral mapping: Synchrotron-based machine learning approach for 2D characterization with coupled micro XRF-XRD”. Computers and Geosciences. 156. July 28, 2021.
Ling, FT, Plattenberger, DA, Peters, CA, Clarens, AF. “Sealing porous media via calcium silicate reactions with CO2 to enhance the security of geologic carbon sequestration”. Environmental Engineering Science. Special Issue: Addressing Society’s Water and Energy Challenges with Reactive Transport Modeling. 38. 3. March 17, 2021.
Hunter, HA, Ling, FT, Peters, CA. “Coprecipitation of Heavy Metals in Calcium Carbonate from Coal Fly Ash Leachate”. Environmental Science & Technology: Water. Nov. 4, 2020. https://dx.doi.org/10.1021/acsestwater.0c00109.
Ling, FT, Post, JE, Heaney, PJ, Santelli, CM, Ilton, ES, Burgos, WD, Rose, AW. “A multi-method characterization of natural terrestrial birnessites”. American Mineralogist. June 4, 2020. https://doi.org/10.2138/am-2020-7303.
Plattenberger, DA, Brown, T, Ling, FT, Lyu, X, Fitts, J, Peters, CA, Clarens, AF. “Feasibility of using reactive silicate particles with temperature-responsive coatings to enhance the security of geologic carbon storage”. International Journal of Greenhouse Gas Control. 95. 102976. April 2020. https://doi.org/10.1016/j.ijggc.2020.102976.
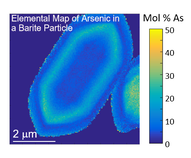
Hunter, HA, Ling, FT, Peters, CA. “Metals Coprecipitation with Barite: Nano-XRF Observation of Enhanced Strontium Incorporation”. Environmental Engineering Science. 37. 4. Nov. 11, 2019. https://doi.org/10.1089/ees.2019.0447.
Plattenberger, DA, Ling, FT, Peters, CA, Clarens, AF. "Targeted Permeability Control in the Subsurface via Calcium Silicate Carbonation". Environmental science & Technology. 53(12). 7136-7144. May 28, 2019. DOI: https://doi.org/10.1021/acs.est.9b00707.
Plattenberger, DA, Ling, FT, Tao, Z, Peters, CA, Clarens, AF. "Calcium Silicate Crystal Structure Impacts Reactivity with CO2 and Precipitate Chemistry". Environmental Science & Technology Letters. 5(9). 558 - 563. Aug. 24, 2018. DOI: https://doi.org/10.1021/acs.estlett.8b00386.
Ling, FT, Hunter, HA, Fitts, JP, Peters, CA, Acerbo, AS, Huang, X, Yan, H, Nazaretski, E, Chu, YS. "Nanospectroscopy Captures Nanoscale Compositional Zonation in Barite Solid Solutions". Scientific Reports. 1 - 11. 8:13041. Aug. 29, 2018. DOI: https://doi.org/10.1038/s41598-018-31335-3.
Ling, FT, Post, JE, Heaney, PJ, Ilton, ES. "The relationship between Mn oxidation state and structure in triclinic and hexagonal birnessites". Chemical Geology. 216 - 217. Feb. 20, 2018. https://doi.org/10.1016/j.chemgeo.2018.01.011.
Ling, FT, Post, JE, Heaney, PJ, Ilton, ES, Kubicki, JD, Santelli, CM. "Fourier-transform infrared spectroscopy (FTIR) analysis of triclinic and hexagonal birnessites". Spectrochimica Acta Part A. 178. 32 - 46. Jan. 17, 2017. https://doi.org/10.1016/j.saa.2017.01.032.
Ilton, E., Post, JE, Heaney, PJ, Ling, FT, Kerisit, SN. "XPS Determination of Mn Oxidation States in Mn(Hydr)oxides". Applied Surface Science. 366. Jan. 11, 2016. https://dx.doi.org/10.1016/j.apsusc.2015.12.159.
Ling, FT, Heaney, PJ, Post, JE, Gao, X. "Transformations from triclinic to hexagonal birnessite at circumneutral pH induced through pH control by common biological buffers". Chemical Geology. 416. Oct. 9, 2015. https://dx.doi.org/10.1016/j.chemgeo.2015.10.007.
Ling, FT, Plattenberger, DA, Peters, CA, Clarens, AF. “Sealing porous media via calcium silicate reactions with CO2 to enhance the security of geologic carbon sequestration”. Environmental Engineering Science. Special Issue: Addressing Society’s Water and Energy Challenges with Reactive Transport Modeling. 38. 3. March 17, 2021.
Hunter, HA, Ling, FT, Peters, CA. “Coprecipitation of Heavy Metals in Calcium Carbonate from Coal Fly Ash Leachate”. Environmental Science & Technology: Water. Nov. 4, 2020. https://dx.doi.org/10.1021/acsestwater.0c00109.
Ling, FT, Post, JE, Heaney, PJ, Santelli, CM, Ilton, ES, Burgos, WD, Rose, AW. “A multi-method characterization of natural terrestrial birnessites”. American Mineralogist. June 4, 2020. https://doi.org/10.2138/am-2020-7303.
Plattenberger, DA, Brown, T, Ling, FT, Lyu, X, Fitts, J, Peters, CA, Clarens, AF. “Feasibility of using reactive silicate particles with temperature-responsive coatings to enhance the security of geologic carbon storage”. International Journal of Greenhouse Gas Control. 95. 102976. April 2020. https://doi.org/10.1016/j.ijggc.2020.102976.
Hunter, HA, Ling, FT, Peters, CA. “Metals Coprecipitation with Barite: Nano-XRF Observation of Enhanced Strontium Incorporation”. Environmental Engineering Science. 37. 4. Nov. 11, 2019. https://doi.org/10.1089/ees.2019.0447.
Plattenberger, DA, Ling, FT, Peters, CA, Clarens, AF. "Targeted Permeability Control in the Subsurface via Calcium Silicate Carbonation". Environmental science & Technology. 53(12). 7136-7144. May 28, 2019. DOI: https://doi.org/10.1021/acs.est.9b00707.
Plattenberger, DA, Ling, FT, Tao, Z, Peters, CA, Clarens, AF. "Calcium Silicate Crystal Structure Impacts Reactivity with CO2 and Precipitate Chemistry". Environmental Science & Technology Letters. 5(9). 558 - 563. Aug. 24, 2018. DOI: https://doi.org/10.1021/acs.estlett.8b00386.
Ling, FT, Hunter, HA, Fitts, JP, Peters, CA, Acerbo, AS, Huang, X, Yan, H, Nazaretski, E, Chu, YS. "Nanospectroscopy Captures Nanoscale Compositional Zonation in Barite Solid Solutions". Scientific Reports. 1 - 11. 8:13041. Aug. 29, 2018. DOI: https://doi.org/10.1038/s41598-018-31335-3.
Ling, FT, Post, JE, Heaney, PJ, Ilton, ES. "The relationship between Mn oxidation state and structure in triclinic and hexagonal birnessites". Chemical Geology. 216 - 217. Feb. 20, 2018. https://doi.org/10.1016/j.chemgeo.2018.01.011.
Ling, FT, Post, JE, Heaney, PJ, Ilton, ES, Kubicki, JD, Santelli, CM. "Fourier-transform infrared spectroscopy (FTIR) analysis of triclinic and hexagonal birnessites". Spectrochimica Acta Part A. 178. 32 - 46. Jan. 17, 2017. https://doi.org/10.1016/j.saa.2017.01.032.
Ilton, E., Post, JE, Heaney, PJ, Ling, FT, Kerisit, SN. "XPS Determination of Mn Oxidation States in Mn(Hydr)oxides". Applied Surface Science. 366. Jan. 11, 2016. https://dx.doi.org/10.1016/j.apsusc.2015.12.159.
Ling, FT, Heaney, PJ, Post, JE, Gao, X. "Transformations from triclinic to hexagonal birnessite at circumneutral pH induced through pH control by common biological buffers". Chemical Geology. 416. Oct. 9, 2015. https://dx.doi.org/10.1016/j.chemgeo.2015.10.007.